Slush Balls
 Close up: typical slush ball dimenstions are 2-3" Photo by Richard Bramwell
Close up: typical slush ball dimenstions are 2-3" Photo by Richard Bramwell
 Slush balls piled up on Ardore Beach with waves rolling under them. The picture looks soutwest toward the Blue Hills ski hills (25 mi). Photo by Richard Bramwell
Slush balls piled up on Ardore Beach with waves rolling under them. The picture looks soutwest toward the Blue Hills ski hills (25 mi). Photo by Richard Bramwell
Red marker is Ardmore Beach on the south end of Georgian Bay, Lake Huron. The blue markers are the Blue Mountain ski hills
Richard Bramwell sent some pictures from January 2011 of an interesting form of ice he found at Ardmore Beach on the south eastern end of Georgian Bay after a night of moderate onshore winds and temperatures around -10˚C. He described them as cobblestone like: rolling around in the water as incoming waves moved under them. I had seen what is probably the same thing a couple times on Lake Champlain at frozen ice edges, far from shore.
 Slush balls on Lake Champlain, frozen into a former ice edge. Scale: Ash skate stiffiner (lower left) about 1" wide.
Slush balls on Lake Champlain, frozen into a former ice edge. Scale: Ash skate stiffiner (lower left) about 1" wide.
After getting Richard's email, I kept watch on several beaches on Lake Champlain until I found them on a very cold (0˚F,-18˚ C), windy (20 mph) day in a small bay just north of the Essex NY ferry dock. The fresh balls are composed entirely of a uniform consistancy slush. It is not as cohesive as water saturated new snow but is more cohesive than fully metamorphosed corn snow slush. Bernard Michel(1) describes slush balls as: "The result of extreme accretion of slush particles. This is produced either by waves along the shores of lakes or in long stretches of very turbulent and wavy flows in rivers."
 Slushballs in a small bay in Essex NY-Lake Champlain, 5 degrees F, January 2011
Slushballs in a small bay in Essex NY-Lake Champlain, 5 degrees F, January 2011
Close up-largest ice balls about 8" long. The light colored bits on the top of some of the balls is drained slush.
The day I found the slush balls on Lake Champlain it was snowing lightly out of the ice fog over the open water on the lake but it did not seem like enough to make this much slush and we did not find slush balls at any of the many other bays and beaches we looked at that day. On the other hand there was plenty of snow falling to nucleate supercooled water on the windblown surface. The low flow from the small stream at the end of the bay also did not seem up to the task of filling the bay with frazil. Also, Ardmore Beach does not have a significant nearby stream and the former ice edge where I had seen them frozen into the ice sheet was far from any streams. Slush particles from unfrozen slush balls between polarizing filters did not give any useful clues that helped make a case for a particular source.
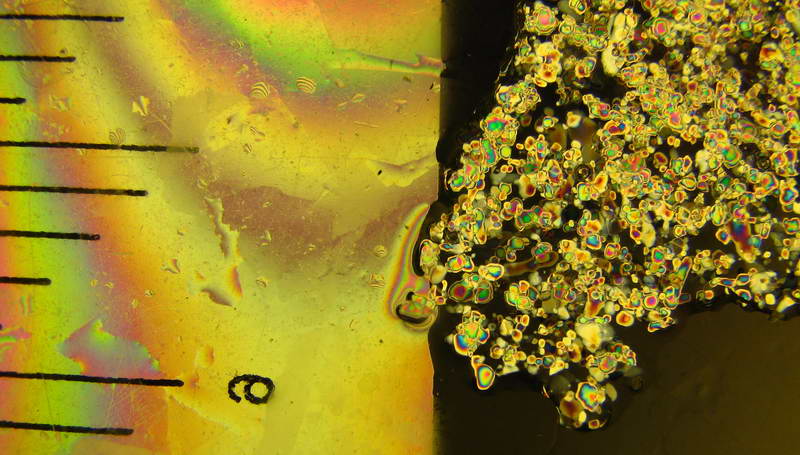 Loose slushball slush between crossed polarizers. Scale increments: 0.1"
Loose slushball slush between crossed polarizers. Scale increments: 0.1"
 Thin section of a frozen slush ball between crossed polarizing filters. The scale is similar to the previous picture.
Thin section of a frozen slush ball between crossed polarizing filters. The scale is similar to the previous picture.
While slush balls have little impact on our use of lake ice they are an interesting curiosity. There are several WWW other references2-3 on the web, one of which was published in 1954.
Bob
********************************************
Update: 1/15/2012
After a 2" snow storm, it has been cold (10 deg F) for the last two days in Burlington. Yesterday we checked a local beach to see if the snow falling into the lake had created to slushballs. There weren't any and it looked like the water was still too warm. Today was another story, there was a band of slushballs about 40 feet wide off the beach. Wind at the time was a few mph and it dropped later in the day. After lunch I came back in my dry suit with lots of clothes underneath. When I waded out to the edge of the slushballs I found evidence for the source and mechanism for making slush as well as got a better understanding of how they evolve. A hypothesis is laid out in the following pictures and captions.
 Slusballs forming off Blodgett Beach in Burlington VT with an onshore breeze of a few mph and temperatures around 10 deg F. You can plainly see the the size of the slushballs gets smaller as you approach the outer edge of the band. It is likely that the balls glom onto each other as they bump into each other. Some of the slushballs are as big as a foot or more.
Slusballs forming off Blodgett Beach in Burlington VT with an onshore breeze of a few mph and temperatures around 10 deg F. You can plainly see the the size of the slushballs gets smaller as you approach the outer edge of the band. It is likely that the balls glom onto each other as they bump into each other. Some of the slushballs are as big as a foot or more. Location close to the pervious picture in a place where the slushball band is wider and after winds have dropped to a couple mph. There is still a one foot swell rolling in which does not roll the slush balls as much and allows them to agglomerate into even bigger balls and to develop frozen top, that freezes into pancake ice on top with 12-16" of slush underneath for the bigger pancakes. The biggest pancakes are easily 2 feet in length.
Location close to the pervious picture in a place where the slushball band is wider and after winds have dropped to a couple mph. There is still a one foot swell rolling in which does not roll the slush balls as much and allows them to agglomerate into even bigger balls and to develop frozen top, that freezes into pancake ice on top with 12-16" of slush underneath for the bigger pancakes. The biggest pancakes are easily 2 feet in length.  The Source: small (1-3") floating, thin (roughly 0.04" or less) dendritc/blades, often with several crystals either tangled with others or growing together. They can be seen in the smooth water separated by a few inches or feet from each other. There has been some lake fog snow falling into the lake in the past hour which might have nucleated these crystals.
The Source: small (1-3") floating, thin (roughly 0.04" or less) dendritc/blades, often with several crystals either tangled with others or growing together. They can be seen in the smooth water separated by a few inches or feet from each other. There has been some lake fog snow falling into the lake in the past hour which might have nucleated these crystals.  Floating crystals 20 feet from the edge of the slush ball deposit. Glove fingers for scale.
Floating crystals 20 feet from the edge of the slush ball deposit. Glove fingers for scale.  Same as previous
Same as previous Same as previous
Same as previous Platy accumulated crystals at the outer edge of the the slush ball band. This platy formation appears to be an accumulation of the thin, flat crystals that are being blown into edge of the band. Waves and impact with existing slush balls break up the these platy sheets into small pieces (slush) on the outer edge of the slushball band. Balls then form if there is sufficient wave action to keep them rolling over. Otherwise the tops freeze and become pancakes. There were smaller (4-10") balls rolling around in the pancake capped slush pieces near the slush formation edge.
Platy accumulated crystals at the outer edge of the the slush ball band. This platy formation appears to be an accumulation of the thin, flat crystals that are being blown into edge of the band. Waves and impact with existing slush balls break up the these platy sheets into small pieces (slush) on the outer edge of the slushball band. Balls then form if there is sufficient wave action to keep them rolling over. Otherwise the tops freeze and become pancakes. There were smaller (4-10") balls rolling around in the pancake capped slush pieces near the slush formation edge.  A look accross the slush band.
A look accross the slush band.
 The same area the next day. It is still 20 deg F but the wind has switched to off shore (south). As you can see most of the slush ball band has departed including parts that were reasonably well attached to the shore. The winds have not been particularly strong in this protected bay but there are some small (few inch) swells being refracted around the points to the west. These swells are creating 'wave break' cracks that cut the attached ice loose.
The same area the next day. It is still 20 deg F but the wind has switched to off shore (south). As you can see most of the slush ball band has departed including parts that were reasonably well attached to the shore. The winds have not been particularly strong in this protected bay but there are some small (few inch) swells being refracted around the points to the west. These swells are creating 'wave break' cracks that cut the attached ice loose.
It looks like a bit more onshore wind speed would have caused smaller, rounder slushballs. The key ingredients for slushball formation seem to be:
- Water in the top few inches of the water near the beach is very close to 32 deg (that is the temperature I measured in a bucket full I brought to shore). A wide band of shallow water might help with this.
- Cold temperatures to supercool the surface water off shore to allow fast growth of dendritic needles. It is the opinion of frazil expert Dr Steve Daley that nucleation takes place at far too little supercooling to be anything other than ice to be the nucleating agent. Snow (even a little), ice fog, frozen water spray in windy condition and broken bits of other crystals are all possible candidates.
- Note: While super cooling of a thin surface layer is taking place off shore to grow the dendritc flake ice/needles I do not believe it plays a role in ice ball formation. When the ice balls are bobbing in the water they have the consistency of a very wet snowball where water/ice particle cohesion holds things together. There is not significant hardening that I would expect would be associated sintereing in supercooled water as is seen in rivers with active frazil whick builds up anchor ice under the water surface.
- A moderate onshore wind that will move the needles growing on the surface downwind but not strong enough to deeply mix the surface water.
- Heavier snow will also contribute to the cooling of the water surface and might contribute significantly to the the slush.
Michel Bernard (1) includes spicules (small or broken needle/dendritic ice pieces) in his definition of frazil. The formation mechanism has some similarities but also significant differences from discoid frazil that commonly forms in turbulent rivers. In River Lake Ice Engineering(4) this is described as needle frazil
Slush, in its various forms, plays an important role in the world of ice. Snow ice, superimposed on the top of an ice sheet often comprises a fair portion of the thickness of many ice sheets by mid/late winter. Discoid frazil ice can develop large ice deposits on rivers that can have significant adverse economic impact. Needle frazil, in addition to making slushballs, is most likely responsible for the large areas of white slush ice that can be often be found mixed with areas of black ice. Needle frazil also accounts for at least some (maybe most) of the small grain (S2) ice found in ice sheets, especially on larger lakes where the needles and flakes get broken up in the early stages of their formation. Snow falling into water can create slush that freezes into spectacular patterns.
Bob
Slightly revised 1/11/14
References:
1) Winter Regime of Rivers and Lakes, Bernard Michel, April 1971, page 3 and 4, Cold Regions Science and Engineering Monograph III-B1a, Cold Regions Research and Engineering Laboratory (CRREL). Note: This is an excellent survey of the practical side of river and lake ice science.
2) Click Here for: A STUDY OF ICE ON AN INLAND LAKE by: James Wilson, James Zumberge and EW Marshall, 1954
3) Click Here from a video taken by Richard Bramwell at Ardmore Beach.
4) River Lake Ice Engineering Edited by G. Ashton, Page 184, Water Resources Publications, LLC, 2010 printing.
5) Detroit Free Press article on Ice Balls
